TIANJIN GNEE BIOTECH CO., LTD., as a professional manufacturer and supplier of Ethyl Acetate (EtOAc, CAS 141-78-6), provides high-quality Ethyl Acetate solvent widely used across coatings, inks, adhesives, pharmaceuticals, and chemical industries. Understanding the chemical properties of Ethyl Acetate helps in selecting the right grade for different Ethyl Acetate industrial uses.
Overview of Ethyl Acetate
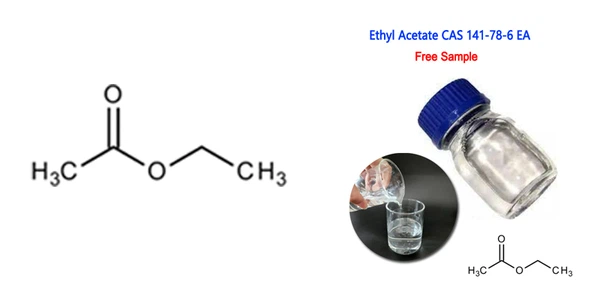
Ethyl Acetate (C4H8O2), also known as ethyl ethanoate or EtOAc, is an important organic ester commonly used as an industrial solvent. It is typically produced through the reaction between ethanol and acetic acid, often in the presence of an acid catalyst.
A simplified reaction is:
CH₃COOH + C₂H₅OH → CH₃COOC₂H₅ + H₂O
This esterification process is reversible, so excess reactants are often used to improve yield in Ethyl Acetate production.
Chemical Structure and Formula
- Chemical Formula: C4H8O2
- Structural Formula: CH₃COOCH₂CH₃
- Molar Mass: 88.11 g/mol
- Functional Group: Ester (-COO-)
Ethyl Acetate is formed when the hydroxyl group of acetic acid is replaced by an ethoxy group, giving it its characteristic properties as a Ethyl Acetate solvent.
Physical Properties of Ethyl Acetate
The physical characteristics of Ethyl Acetate (CAS 141-78-6) contribute to its widespread use:
- Boiling Point: ~77°C
- Melting Point: ~ -83°C
- Density: ~0.89 g/cm³
- Solubility: Slightly soluble in water, highly soluble in most organic solvents
Due to its fast evaporation and good solvency, it is widely used as Ethyl Acetate solvent for coatings and inks and in Ethyl Acetate industrial solvent applications.
Chemical Properties of Ethyl Acetate
Hydrolysis Reaction
Ethyl Acetate can react with water in the presence of acid or base catalysts:
- Produces ethanol and acetic acid
- Commonly used in controlled hydrolysis processes
This reaction is widely studied in Ethyl Acetate chemical reactions and industrial chemistry.
Transesterification
Another important reaction is transesterification:
- The ethoxy group in Ethyl Acetate (EtOAc) can be replaced by another alcohol group
- Produces different ester compounds
- Useful in chemical synthesis and industrial applications
Ester Stability and Reactivity
As an ester compound, Ethyl Acetate:
- Is relatively stable under normal conditions
- Can undergo reactions under acidic or basic environments
- Serves as a reactive intermediate in organic synthesis
Industrial Significance of Ethyl Acetate
Because of its chemical properties, Ethyl Acetate (CAS 141-78-6) is widely used in:
- Coatings and paints: As a fast-evaporating solvent
- Adhesives: Enhancing bonding performance
- Printing inks: Improving drying efficiency
- Pharmaceuticals: Used as a Ethyl Acetate pharmaceutical extraction solvent
- Food industry: Applied as food grade Ethyl Acetate for flavoring and extraction
These diverse applications highlight the importance of Ethyl Acetate industrial uses.
Production Method
Ethyl Acetate is commonly produced via:
- Esterification of ethanol and acetic acid
- Catalyzed by acids such as sulfuric acid
- Industrial distillation and purification processes
This method ensures consistent quality of Ethyl Acetate (C4H8O2) for large-scale applications.

Conclusion
Ethyl Acetate (EtOAc, CAS 141-78-6) is a versatile ester with well-defined chemical and physical properties, including reactivity through hydrolysis and transesterification. These characteristics make it an essential Ethyl Acetate solvent used across coatings, adhesives, inks, pharmaceuticals, and other industries.
As a trusted Ethyl Acetate manufacturer and supplier, TIANJIN GNEE BIOTECH CO., LTD. ensures stable quality and reliable supply of Ethyl Acetate (C4H8O2) to meet global industrial demands.
Contact us today for bulk supply and technical support for Ethyl Acetate (EtOAc).





